Iso 13485 Free Course
Iso 13485 Free Course - Whether you’re looking to learn more about a new standard, or you’re new to the world of standards altogether, our free introductory courses will help you gain the essentials. Upon completion of this course, you will be able to: Choose from a range of free courses to enhance your knowledge of iso standards. This free online iso 13485 certification training course will teach you about quality management systems for medical devices. Learn all you need to know about a medical device qms in our iso 13485 foundations course for beginners. You will need eight hours to complete the iso 13485 foundations course, 15 hours for the iso 13485 internal auditor course, and 20 hours for the iso 13485 lead auditor or lead implementer course. It means decisions are made free from any engagements of influences which could affect the objectivity of decision making. This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001:2015 and also it serves to facilitate global alignment for medical device quality management systems. This course is designed to provide you with a thorough foundation in the essential aspects of iso 13485:2016 standards, equipping you with the knowledge and skills needed to excel in quality management for medical devices. This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with. This free online iso 13485 certification training course will teach you about quality management systems for medical devices. Iso 13485 specifies qms requirements for the medical device manufacturing industry. Understand why iso 13485 quality management systems are important; Alison offers free online engineering iso courses. Our free resources pages also provide information on how you can engage our experts, when needed. If you’ve ever struggled to find iso 13485 training courses, webinars, online webinars and training materials like powerpoint presentations, this is a great site for you. Darüber hinaus lernen die teilnehmer mehr über die beziehung zwischen iso 13485 und iso 14971 „anwendung des risikomanagements auf medizinprodukte“. This free online iso 13485 certification training course will teach you about quality management systems for medical devices. All of the key iso 13485 and eu mdr documents, records, and templates necessary to complete your certification process. This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with. This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001:2015 and also it serves to facilitate global alignment for medical device quality management systems. Learn online for iso 9001, iso 27001, iso 14001, iso45001 and more. Describe why iso 13485 is important in helping ensure. Alison offers free online engineering iso courses. This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with. You will study the iso 13485:2016 standard, learn how it was developed, and look into the practical steps to company certification on iso 13485. This training course explores the requirements. Enroll for free iso 9001 lead auditor Find iso 13485 training and lead auditor classes at asq.org. Our free resources pages also provide information on how you can engage our experts, when needed. This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001:2015 and also. Recognize the benefits of an iso13485 qms for your organisation; If you’ve ever struggled to find iso 13485 training courses, webinars, online webinars and training materials like powerpoint presentations, this is a great site for you. This course is designed to provide you with a thorough foundation in the essential aspects of iso 13485:2016 standards, equipping you with the knowledge. Gain vital understanding of internationally recognized standards in engineering. Learn online for iso 9001, iso 27001, iso 14001, iso45001 and more. Enter your details to gain access to our free iso training courses. Free resources for iso 13485 implementation, auditing and training including tips on documentation requirements, required records, key links and articles, and more. Learn all you need to. You will need eight hours to complete the iso 13485 foundations course, 15 hours for the iso 13485 internal auditor course, and 20 hours for the iso 13485 lead auditor or lead implementer course. Get started on your certification journey now. This free iso 13485 lead implementer course will teach you how to become an independent consultant or practitioner for. Whether you’re looking to learn more about a new standard, or you’re new to the world of standards altogether, our free introductory courses will help you gain the essentials. Find iso 13485 training and lead auditor classes at asq.org. Upon completion of this course, you will be able to: This free iso 13485 lead implementer course will teach you how. Recognize the benefits of an iso13485 qms for your organisation; If you're looking to simply learn the quality management system requirements or become a certified internal or lead auditor, training with our experts will provide what you need. This course is designed to provide you with a thorough foundation in the essential aspects of iso 13485:2016 standards, equipping you with. Enroll for free iso 9001 lead auditor Free resources for iso 13485 implementation, auditing and training including tips on documentation requirements, required records, key links and articles, and more. Enter your details to gain access to our free iso training courses. Whether you’re looking to learn more about a new standard, or you’re new to the world of standards altogether,. This elearning training course offers insight into the use of iso 13485:2016 as the basis for a quality management system (qms) implemented by medical device organizations. Iso 13485 is the cornerstone of quality management systems (qms) for medical devices. Our free resources pages also provide information on how you can engage our experts, when needed. Darüber hinaus lernen die teilnehmer. Learn all you need to know about a medical device qms in our iso 13485 foundations course for beginners. Whether you’re looking to learn more about a new standard, or you’re new to the world of standards altogether, our free introductory courses will help you gain the essentials. This course is designed to provide you with a thorough foundation in the essential aspects of iso 13485:2016 standards, equipping you with the knowledge and skills needed to excel in quality management for medical devices. This course teaches all you need to know about iso 13485, helping you become an independent consultant for the standard. Find iso 13485 training and lead auditor classes at asq.org. This elearning training course offers insight into the use of iso 13485:2016 as the basis for a quality management system (qms) implemented by medical device organizations. You will study the iso 13485:2016 standard, learn how it was developed, and look into the practical steps to company certification on iso 13485. Describe why iso 13485 is important in helping ensure patient safety This free online iso 13485 certification training course will teach you about quality management systems for medical devices. Appreciate how your role is relevant to the iso13485 qms; It means decisions are made free from any engagements of influences which could affect the objectivity of decision making. Iso 13485 is the cornerstone of quality management systems (qms) for medical devices. Gain vital understanding of internationally recognized standards in engineering. Upon completion of this course, you will be able to: This training course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with. World’s most popular iso 13485 courses for beginners, and for experienced professionals.Online Quality Management for Medical Devices and ISO 13485 Course
Free download of ISO 13485 & other medical device standards
ISO 13485 Implementation Course JC Auditors Academy
Free ISO 13485 Foundations Online Course Advisera Training
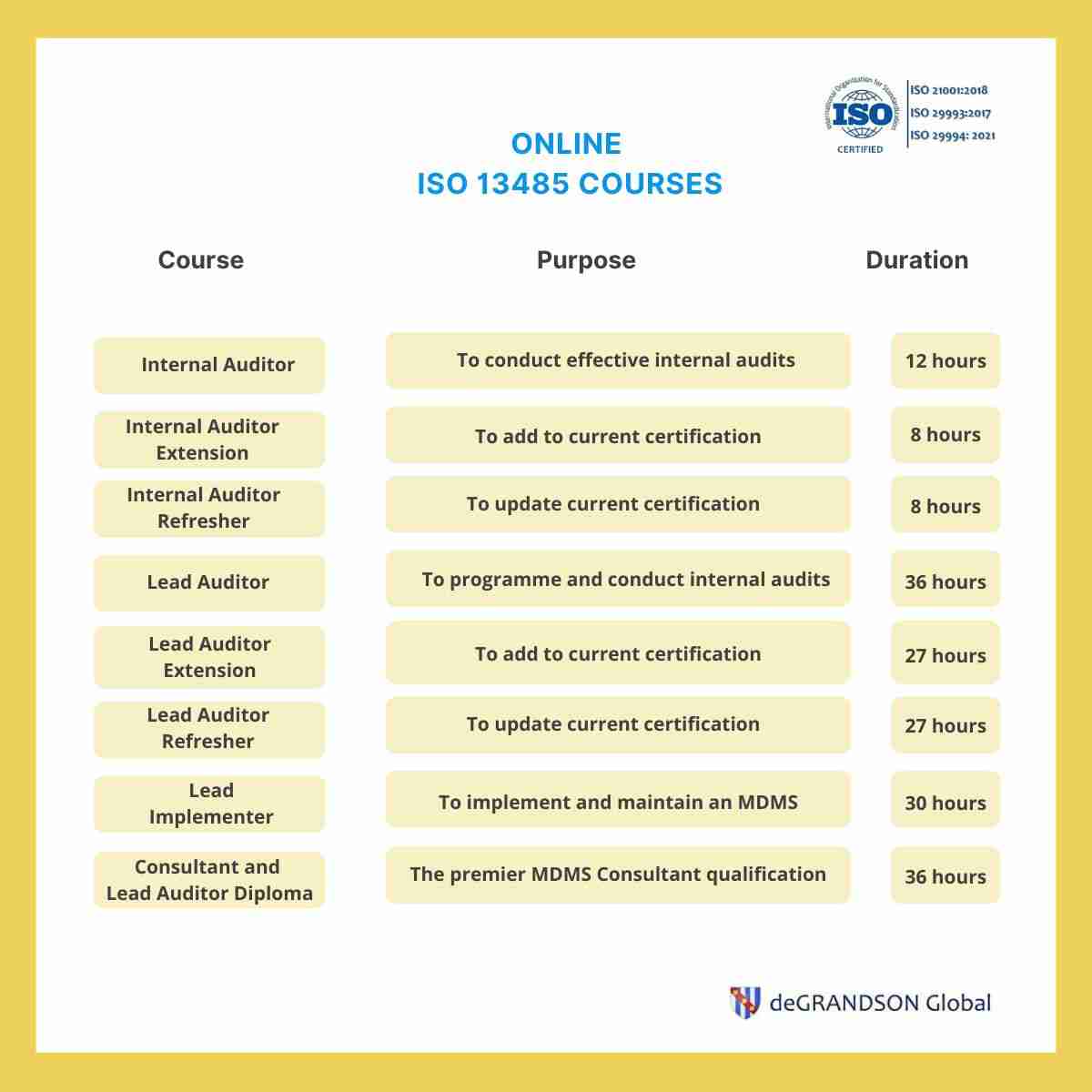
ISO 13485 Online Training available 24/7 ISO 21001 accredited
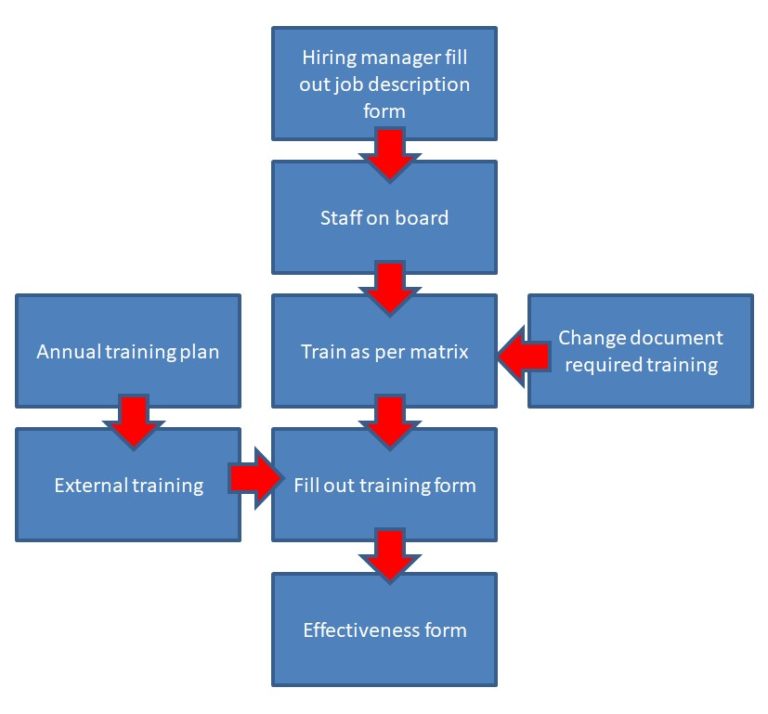
Free ISO 13485 Training Procedure Template
ISO 13485 Certification Training Courses
ISO 13485 Online Training available 24/7 ISO 21001 accredited
Free ISO 13485 Foundations Online Course Advisera Training
Free ISO 13485 Foundations Online Course Advisera Training
You Will Need Eight Hours To Complete The Iso 13485 Foundations Course, 15 Hours For The Iso 13485 Internal Auditor Course, And 20 Hours For The Iso 13485 Lead Auditor Or Lead Implementer Course.
If You're Looking To Simply Learn The Quality Management System Requirements Or Become A Certified Internal Or Lead Auditor, Training With Our Experts Will Provide What You Need.
Choose From A Range Of Free Courses To Enhance Your Knowledge Of Iso Standards.
Understand Why Iso 13485 Quality Management Systems Are Important;
Related Post: